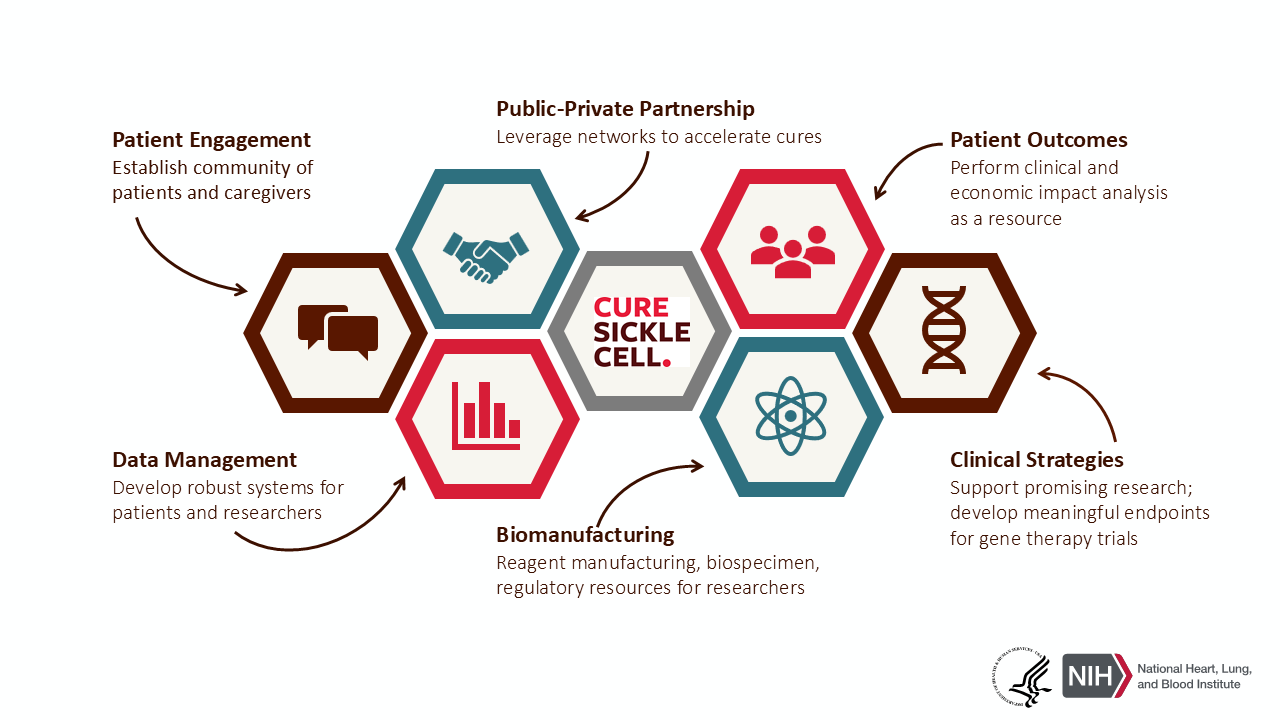
CureSCi and collaborators have addressed critical areas of need established before the launch, including:
Advancing Gene Therapy Clinical Trials
Gene therapy clinical trials are the bridge between innovation and real-world impact, helping to bring the most promising treatment options to those who need them the most. Several key foundational activities played a pivotal role in advancing CureSCi gene therapy clinical trials, including working groups focused on:
- Assay development: to ensure the design, creation, and utilization of tests to measure, evaluate, and confirm the efficacy of gene therapies.
- Manufacturing: to develop a process/oversight to confirm that gene therapies are produced with consistent quality and avoid variability in processes that could affect outcomes.
- Data Management: to identify, document, collect, and harmonize SCD Common Data Elements (CDEs) to support secondary analyses and future clinical trials.
Learn about CureSCi clinical trials, by visiting the Clinical Trials Page.
Find information about CureSCi data standards and tools here.
